
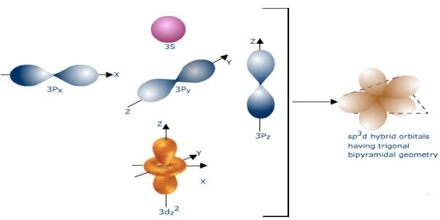
One s and one p atomic orbital are mixed in sp hybridization, one s and 2 p atomic orbitals are mixed in sp² hybridization, and one s and 3 p atomic orbitals are mixed in sp³ hybridization. The two electrons previously in the s orbital have been dispersed to the two half-filled sp orbitals. 
The sp set consists of two similar orbitals that are 180 degrees apart.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


 RSS Feed
RSS Feed
